 
In molecules with more than three atoms, there are many more possible geometries. However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. Predicting Electron Pair Geometry and Molecular Structure. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. The VSEPR chart given above confirms that molecules or molecular ions with an AX 4 generic formula have an identical electron geometry and molecular geometry or shape, i.e. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. 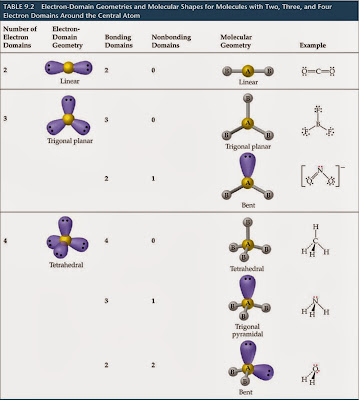
Total number of valence electrons in N2H2 52 + 12 12. Hydrogen belongs to group 1 and has 1 valence electron. Nitrogen belongs to group 15 and has 5 valence electrons. Notice that there are two kinds of electron groups in this structure. We can draw the Lewis structure on a sheet of paper. Lets consider the Lewis structure for CCl 4. A polyatomic molecule contains more than two atoms. In the case of N2H2, a single molecule has two atoms of nitrogen and two atoms of hydrogen. For bent molecular geometry when the electron-pair geometry is tetrahedral the bond angle is around 105 degrees.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
